Trusted by Biologics Teams Building the Next Generation of Therapies





One Platform. Every Stage of Biologics R&D.
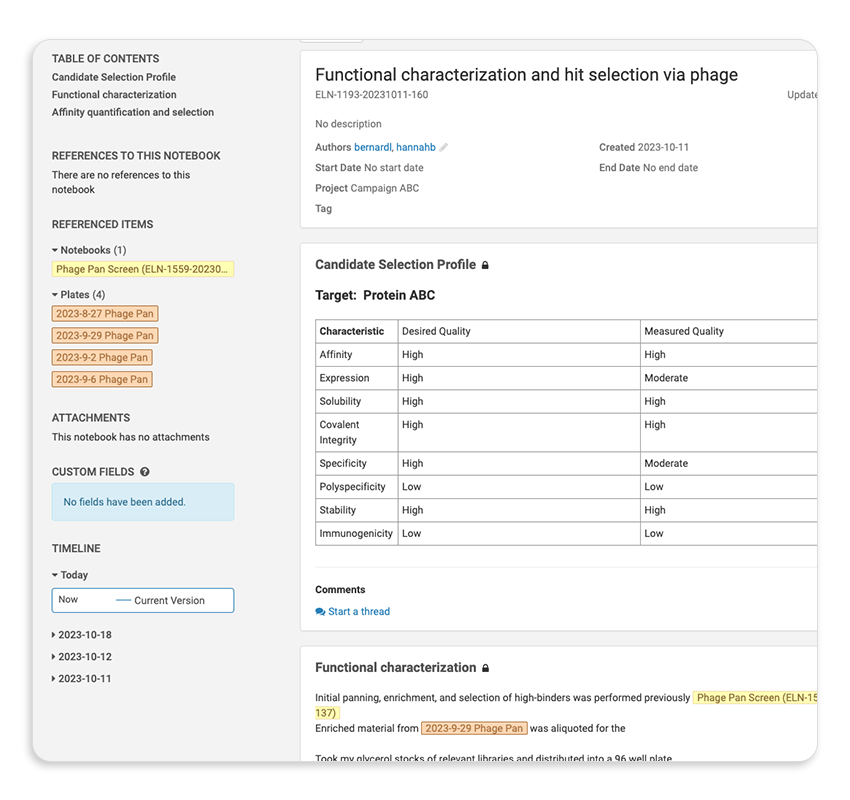
Take a TourMaintain Full Sample and Entity Lineage
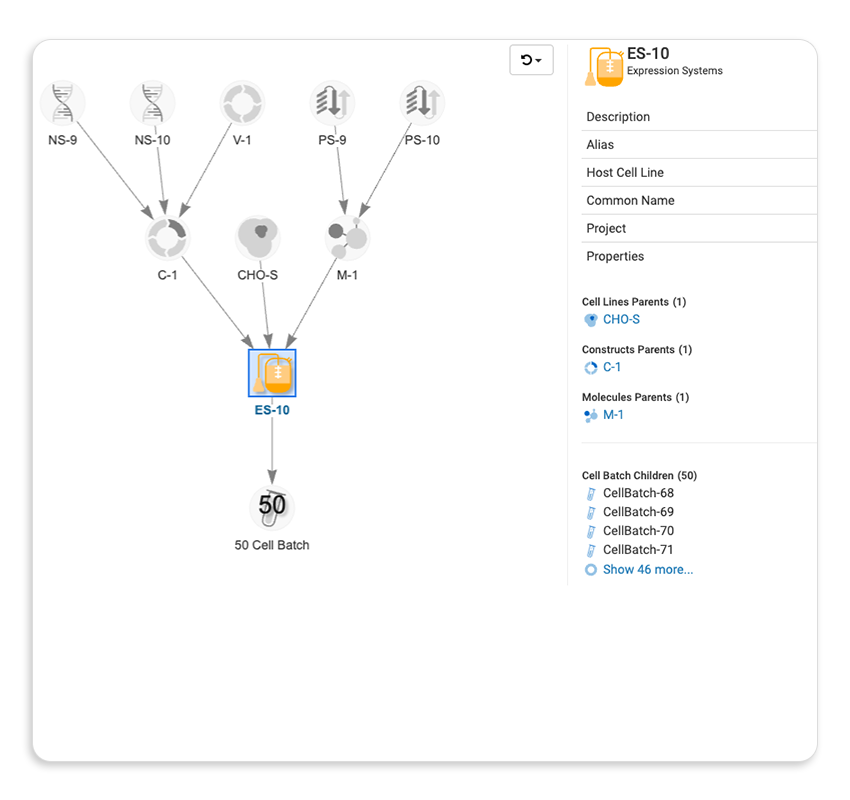
Track every protein, construct, cell line, and assay result back to its origin, across programs, teams, and stages.
Eliminate Manual Data Reconciliation
Replace fragmented spreadsheets and siloed ELNs with structured, connected data that scientists can actually trust.
Accelerate Decisions Across Programs
Move faster by easily surfacing assay results, screening outcomes, and entity relationships.
Collaborate Across Functions and CROs
Give internal R&D teams and external partners shared access to clean, consistent data.
Enforce Data Standards Without IT Overhead
Configurable workflows, validation rules, and role-based access keep data quality high without requiring IT support.
Build a Foundation for AI and Analytics
Structured, audit-ready datasets so your data is ready for analytics, AI/ML, and regulatory review.
See How LabKey Handles the Complexity Other LIMS Can't
Unlike rigid LIMS systems or general-purpose ELNs, LabKey Biologics LIMS is designed from the ground up for protein-based R&D- configurable to your workflows, extensible as your science evolves, and built to give scientists control.
Start Tour
Biologics LIMS Pricing |
Biologics LIMSIdeal for growing biotechs focused on antibody discovery. $495/user/month*Take a Tour |
|||
|---|---|---|---|---|
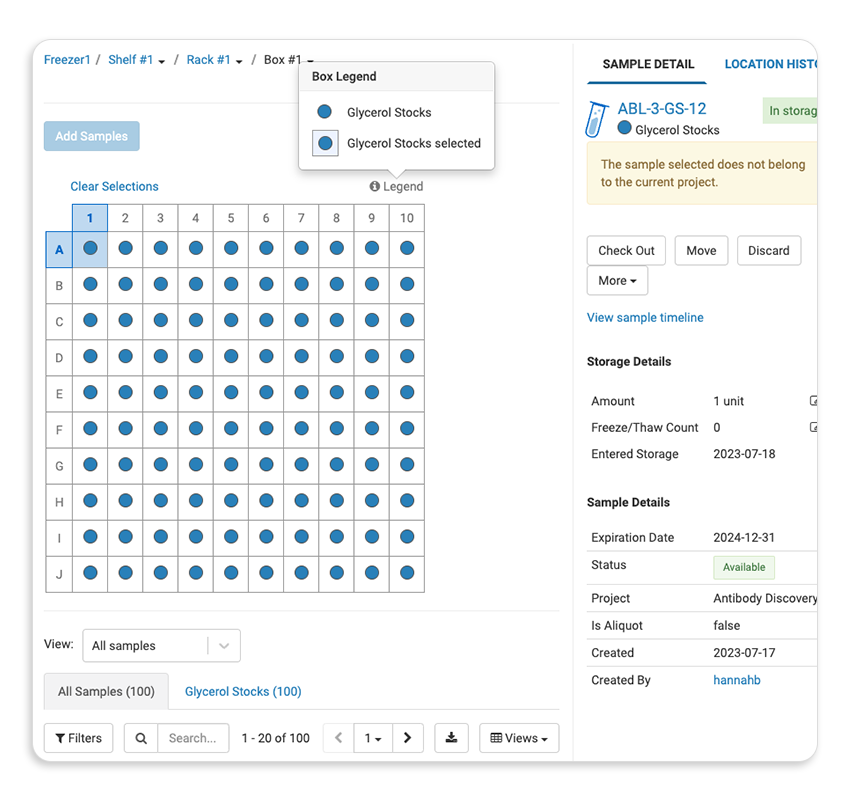
| Sample/Freezer Management |  |
|||
| Electronic Lab Notebook |  |
|||
|
Biological Entity Registration

|
 |
|||
|
Sequence Registration & Annotation

Nucleotide and protein sequence registration, annotation and visualization for all sequence parts. |
 |
|||
|
Compound Registration

Including physical property calculations and SMILES support. |
 |
|||
| Sample & Entity Lineage Tracking |  |
|||
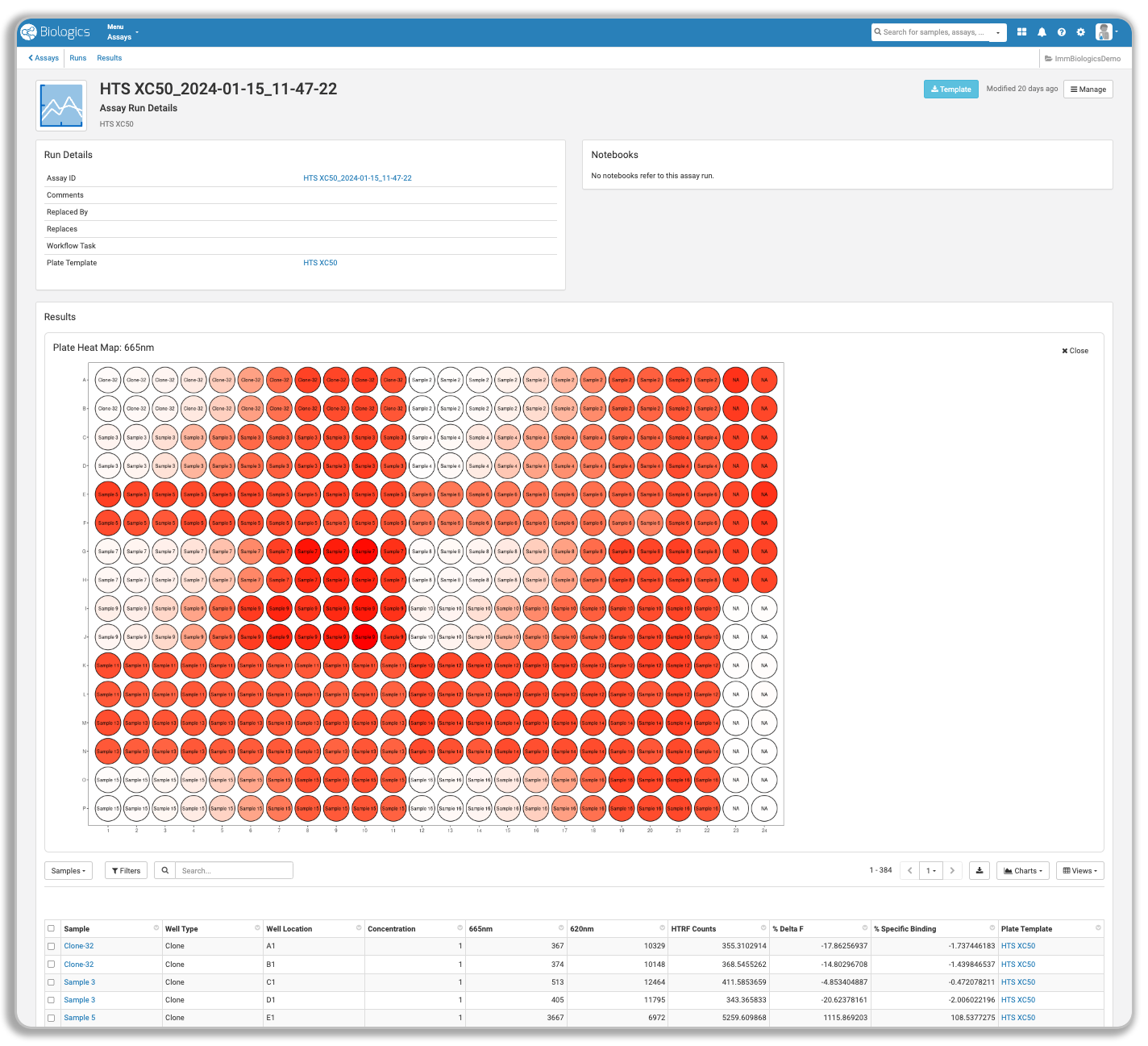
| Assay Data Management |  |
|||
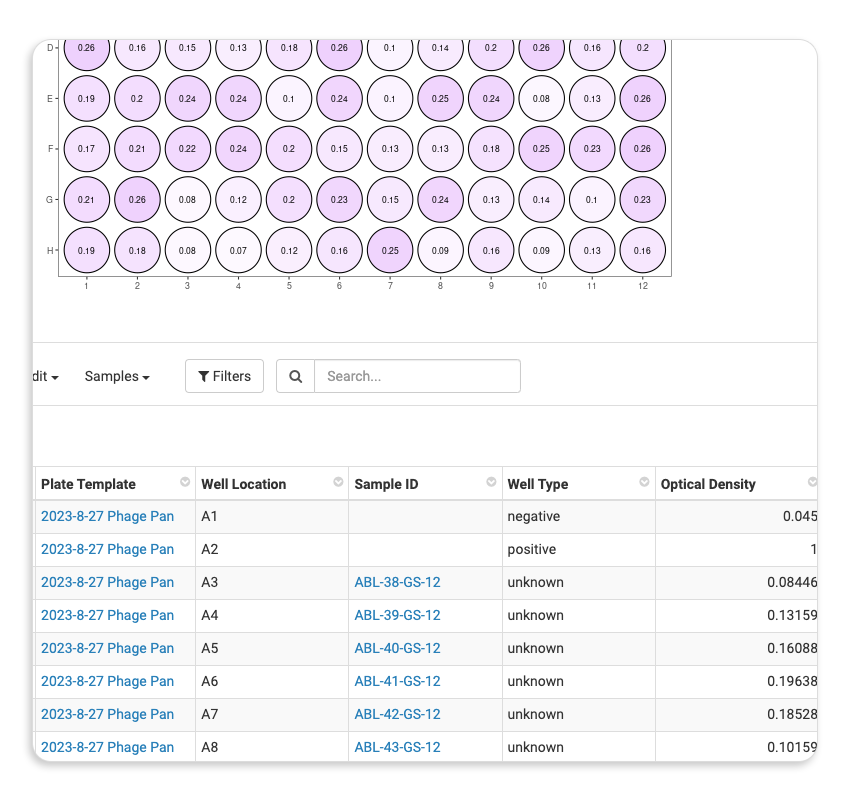
| Plate Management |  |
|||
| Work Request Management |  |
|||
| API Access |  |
|||
| Secure Cloud Hosting |  |
|||
| Audit Trail & 21 CFR Part 11 Compliant |  |
|||
|
Analytics Integrations

Spotfire, Tableau, R/R Studio/R Workbench / Rserve and more. |
 |
|||
|
Database Integrations

Amazon S3, PostgreSQL, MS SQL Server, SAS, Oracle, MySQL, Amazon Redshift |
 |
|||
| Automated Data Capture/Transfer |  |
|||
| Sample & Assay QC |  |
|||
| Media Registration |  |
|||
|
Technical Support for Extensibility

Support for technical extension, customization, and systems integration. |
 |
|||
| On-premise Installation | add-on | |||
| Software Validation | add-on | |||
| Migration Services | add-on | |||
| *Pricing for a minimum of 10 users, billed annually. Volume discount pricing is available. | ||||
Learn more about our LIMS software for biotechs.
Biologics LIMS Frequently Asked Questions
A Biologics LIMS is a laboratory information management system designed specifically for protein-based R&D — antibody discovery, cell line development, construct design, and characterization workflows. General-purpose LIMS platforms and chemical LIMS adapted for biologics weren't built to handle the complex, branching relationships between molecules, variants, constructs, cell lines, samples, and assay results. A purpose-built system provides native data models for all of it.
Most LIMS platforms were built around chemical or clinical workflows and adapted for biologics after the fact. LabKey Biologics LIMS was designed from the ground up for protein R&D — with native data models for antibodies, antigens, vectors, cell lines, and peptides, and a connected architecture that maintains lineage from construct design through characterization. Your team configures it to your science rather than working around a system that wasn't built for it.
A Bioregistry is a centralized system for registering and managing every biological entity your programs depend on — antibodies, antigens, peptides, vectors, cell lines, and more — along with the relationships between them. As programs scale, tracking constructs and variants across disconnected tools creates errors and delays. LabKey's integrated Bioregistry keeps every entity linked to its origin, related samples, and associated assay data, so nothing gets lost as programs grow.
Yes. LabKey Biologics LIMS gives internal R&D teams and external partners like CROs shared access to clean, consistent data through role-based access controls. External collaborators can contribute data without compromising governance, and every action is logged and traceable. And, data quality holds whether work is happening in-house or externally.
Yes. LabKey Biologics LIMS includes a full audit trail and is 21 CFR Part 11 compliant, with role-based access controls, timestamped records of every user action, and data validation rules built into the platform. These capabilities support regulatory alignment without requiring separate compliance tooling.
LabKey captures assay results in a standardized, queryable framework that works across assay types and protein classes — connected directly to the samples and entities that generated them. Your team can query across experiments, visualize performance trends, and surface top candidates without manually reconciling data from separate files or systems.
Yes. LabKey Biologics LIMS supports integrations with analytics platforms including Spotfire, Tableau and R/R Studio, as well as database connections to Amazon S3, PostgreSQL, MS SQL Server, SAS, Oracle, MySQL, and Amazon Redshift. Structured, audit-ready datasets mean your data is ready for analytics, AI/ML workflows, and regulatory review without additional cleanup.